-

Product Information
Scientific name: MWTX-003
Brand name: Ν/Α
RESPONSIBLE: Disc Medicine Inc. and Mabwell Therapeutics -

Clinical Trial/Study Information
Trial Name: N/A
Code: NCT06050915
Phase: 1 (Proof-of-Mechanism)
Eligible patient diagnosis: Healthy Volunteers
No. of Patients enrolled: 64 [anticipated]
Study Sites: 1 Sites per countryAnticipated completion date: 2024
Scope of the Study / Aim: Targets TMPRSS6 with an antibody to enable the increase of hepcidin and reduction of iron -

Clinical Trial/Study Information
Trial Name: N/A
Code: NCT07187973
Phase: 1b
Eligible patient diagnosis: Sickle Cell (HbSC or HbSS); age 18 years and older
No. of Patients enrolled: 24 (anticipated) (Last update: 21/12/2025)
Study Sites: 1 Sites per countryAnticipated completion date: 2027
Scope of the Study / Aim: Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of DISC-3405 in Participants with Sickle Cell Disease
Update: 31 March 2026
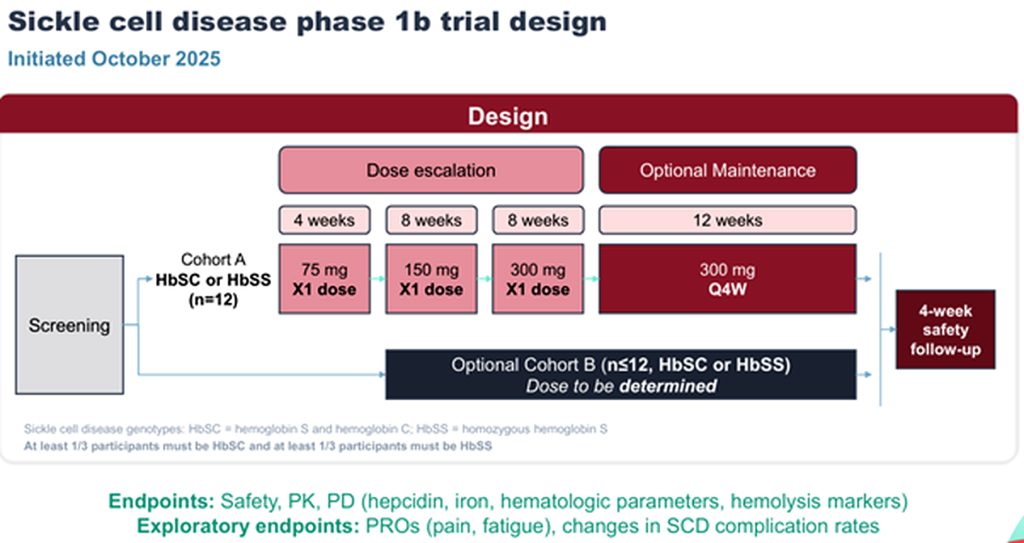
Source: Corporate Presentation, March 2026
Update: 22 December 2025
- Progressing the ongoing Phase 2 study of DISC-3405 in patients with polycythemia vera, with initial data expected in 2026.
- Initiated Phase 1b study of DISC-3405 in patients with SCD in October 2025, with initial data expected in 2026.
Sources: ASH 2025 Corporate Presentation
Disc Medicine Reports Third Quarter 2025 Financial Results and Provides Business Update – Disc Medicine Inc.
Disc Medicine Provides Update on Hematology Portfolio and Outlines Near-Term Business Objectives and Anticipated Milestones – Disc Medicine Inc.
Update: 30 September 2025
Phase 2 study in polycythemia vera initiated; initial data expected in 2026.
Update: 30 June 2025
- Data presented at the 30th EHA Annual Congress (12 – 15 June) in Milan, Italy, showed that DISC-3405 was well tolerated with no serious adverse events in a phase 1 study with healthy volunteers. Increases in hepcidin were observed as well as reductions in serum iron, supporting a once-a-month dosing regimen.
- This data further supports initiating a phase 2 study in polycythemia vera.
Update: 31 March 2025
- FDA has granted orphan drug designation to DISC-3405 for polycythemia vera
- Phase 2 study in polycythemia vera expected to start in the first half of 2025
- Preclinical data in sickle cell disease highlight the potential of DISC-3405 to benefit SCD by restricting iron
Sources: https://ir.discmedicine.com/news-releases/news-release-details/disc-medicine-reports-fourth-quarter-and-full-year-2024
https://www.pharmaceutical-technology.com/news/disc-wins-orphan-drug-tag-for-rare-blood-cancer/
https://ir.discmedicine.com/static-files/28eb4a9e-383f-4605-b904-b7cdecc2d19b
Update: 19 December 2024
New data
- Data presented at the 66th ASH Annual Congress (7-10 December 2024) in San Diego (USA):
- Preliminary findings in healthy volunteers in both single ascending dose (SAD) and multiple ascending dose (MAD) groups showed meaningful and sustained serum iron reductions, a safe and tolerable profile, and no serious adverse events.
- These results support Disc’s plans to advance the DISC-3405 program into a Phase 2 study in polycythemia vera in 2025.
Source: Phase 1 Healthy Volunteer Study of DISC-3405, a Recombinant Humanized Antibody Targeting TMPRSS6
https://ir.discmedicine.com/news-releases/news-release-details/disc-medicine-presents-positive-clinical-and-translational-data
Update: 30 September 2024
- Data from multiple ascending doses is expected by the end of the year.
Update: 30 June 2024
- Data presented at the 29th EHA Annual Congress (13 – 16 June 2024) in Madrid (Spain) showed that the Phase 1 trial in healthy volunteers demonstrated >50% serum iron suppression and sustained hepcidin induction, thus demonstrating clinical proof-of-mechanism as an iron restriction agent.
Update: 31 March 2024
No update available.
Update: 20 December 2023
DISC-3405 (formerly MWTX-003) was in-licensed from Mabwell Therapeutics. It received IND approval from the US FDA in November 2022 and was fast-tracked by the FDA for polycythemia vera in September 2023.
Preclinical studies of DISC-3405 have demonstrated an increase in hepcidin production and suppression of serum iron levels in animal models of beta-thalassemia and polycythemia vera.
A phase 1 clinical study in healthy volunteers in October 2023 was initiated.
DISC-3405 could potentially treat multiple iron overload disorders, such a polycythemia vera, beta thalassemia, and other indications.
By inhibiting TMPRSS6, DISC-3405, an anti-TMPRSS6 antibody, is designed to increase the production of hepcidin to therapeutically reduce iron.
Sources: https://www.discmedicine.com/our-pipeline/mat-2-inhibitor/
https://ir.discmedicine.com/news-releases/news-release-details/disc-medicine-reports-third-quarter-2023-financial-results-and
https://ir.discmedicine.com/news-releases/news-release-details/disc-medicine-initiates-phase-1-study-disc-3405-anti-tmprss6-mab
Update: 30 September 2023
No update available.
Update: 30 June 2023
Phase 1 trial to begin in the second half of 2023 with a focus on polycythemia vera to prove the therapeutic hypothesis and assess safety. Subsequently, Phase 1b proof-of-concept studies can be undertaken in thalassaemia.
Source: https://ir.discmedicine.com/static-files/549caf12-e7be-45ff-8667-86908e4e6bdd
Update: 31 March 2023
- Disc Medicine Inc and Mabwell Therapeutics have entered into an agreement to obtain an exclusive license to a portfolio of monoclonal antibodies targeting TMPRSS6 (Transmembrane Serine Protease 6, also known as Matriptase-2) including the phase 1-ready drug candidate MWTX-003.
- Disc plans to initiate a phase 1 trial in healthy volunteers in the second half of 2023. MWTX-003 has the potential to address a wide range of hematologic disorders including polycythemia vera and beta-thalassemia by controlling iron homeostasis. Genetic studies show that TMPRSS6 affects red blood cell formation by controlling the level of iron that is available for erythropoiesis.
- Clinical and non-clinical evidence has shown that increasing hepcidin and reducing iron levels by inhibiting TMPRSS6 has potential to treat hematologic disorders.
- MWTX-003 is phase 1-ready and received acceptance of an Investigational New Drug (IND) application from the U.S. Food and Drug Administration (FDA) in November 2022.
Sources: https://www.globenewswire.com/news-release/2023/01/20/2592372/0/en/Disc-Medicine-Announces-Exclusive-Licensing-Agreement-with-Mabwell-Therapeutics-for-Novel-Anti-TMPRSS6-Monoclonal-Antibodies-to-Modulate-Iron-Homeostasis.html
https://ir.discmedicine.com/news-releases/news-release-details/disc-medicine-reports-first-quarter-2023-financial-results-and






