News
-

Applications now open for TIF’s Renzo Galanello Fellowship Programme 2020
TIF‘s Renzo Galanello Fellowship Programme is back again in 2020! A small number of physicians, specialists in the field of haematology, paediatrics or internal medicine, will have the opportunity…
Read More » -

‘FAiTh – Fight Against Thalassemia Association’ presents its activities for January 2020
The ‘FAiTh – Fight Against Thalassemia’ group, a longtime member of TIF operating in Pakistan, is dedicated to support, serve and love people with thalassaemia and their families, intending…
Read More » -

FDA grants Orphan Drug Designation for ARU-1801 for the Treatment of Sickle Cell Disease & β-thalassaemia
An investigational gene therapy for Sickle Cell Disease (SCD) and β-thalassaemia, known as ARU-1801, has been given Orphan Drug Designation an orphan drug by the U.S. Food and Drug Administration (FDA), a…
Read More » -

Chiesi Group and Apotex Inc. finalize agreement for the acquisition of Ferriprox® (Deferiprone)
Chiesi Farmaceutici and Apotex (together with its American subsidiary, ApoPharma Usa), a company engaged in haematology and in the area of neurodegenerative diseases, have announced that they have signed…
Read More » -

TIF’s latest Newletter is out now!
Catch up with our most recent newsletter and get informed about TIF’S activities and achievements in the past few months! If you would also like to receive our news directly…
Read More » -

Bluebird bio announces 1st commercial launch of ZYNTEGLO™in Germany
Bluebird bio, Inc. announced earlier today the launch of ZYNTEGLO™ in Germany, the company’s one-time gene therapy for patients 12 years and older with transfusion-dependent β-thalassaemia (TDT) who do…
Read More » -

Deferasirox Accord: A new generic of Exjade recommended for approval by the EMA
The Committee for Medicinal Products for Human Use (CHMP), the European Medicines Agency‘ (EMA) committee responsible for human medicines, has recently adopted a positive opinion recommending the granting of a…
Read More » -
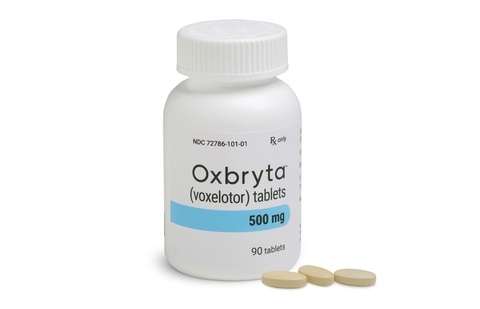
FDA Approves Oxbryta™ (Voxelotor), the First Medicine Specifically Targeting the Root Cause of Sickle Cell Disease
Global Blood Therapeutics, Inc. announced only a few hours ago that the U.S. Food and Drug Administration (FDA) has granted accelerated approval for Oxbryta™ (previously known as Voxelotor) tablets…
Read More » -

Iranian Blood Transfusion Organization (IBTO) Newsletter – November 2019
The latest issue from the Iranian Blood Transfusion Organization’s (IBTO) newsletter is now available on TIF’s website!
Read More » -



